- Blog
- 2d rpg games steam
- Adobe digital editions 4-5 mobile app
- Modelsim verilog
- Zbrush 4r8 crack download
- Prithviraj chauhan delhi
- Kanaa movie review
- Best dolby atmos movies blu ray
- Free word search puzzle maker with clues
- Depriester chart for equilibrium
- Maple 2019
- Turkey is a healthy meat the office script
- My candy love cheats kizmet
- Ms clean kit threading
- Sonicwall netextender or mobile connect
- Xcom 2 star wars total conversion nexus
- Vinylmaster pro drop shadow reverse
- Unattach games from wargaming game center
- Antichrist movie nude scenes
- Printopia 2-1-8 -zip
- Synfig studio console
- Key for windows movie maker 2019
- Crpf pay slip print
- Arduino ide esp8266 firmware
- Protonvpn refund
- Premier aircraft design b1900d
- Blog
- 2d rpg games steam
- Adobe digital editions 4-5 mobile app
- Modelsim verilog
- Zbrush 4r8 crack download
- Prithviraj chauhan delhi
- Kanaa movie review
- Best dolby atmos movies blu ray
- Free word search puzzle maker with clues
- Depriester chart for equilibrium
- Maple 2019
- Turkey is a healthy meat the office script
- My candy love cheats kizmet
- Ms clean kit threading
- Sonicwall netextender or mobile connect
- Xcom 2 star wars total conversion nexus
- Vinylmaster pro drop shadow reverse
- Unattach games from wargaming game center
- Antichrist movie nude scenes
- Printopia 2-1-8 -zip
- Synfig studio console
- Key for windows movie maker 2019
- Crpf pay slip print
- Arduino ide esp8266 firmware
- Protonvpn refund
- Premier aircraft design b1900d
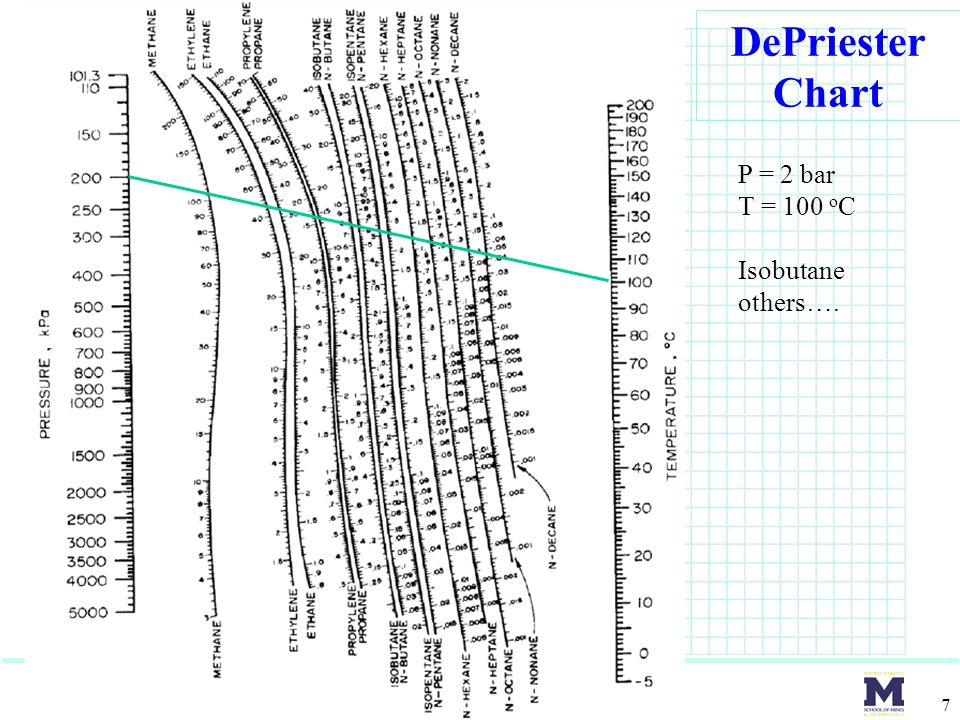
- Depriester chart for equilibrium software#
- Depriester chart for equilibrium license#
- Depriester chart for equilibrium tv#
Pure vaporįor an ideal gas, Equation 6 holds true. Kideal is a function of system temperature and pressure and independent of composition. fiL and fiV are the fugacities of component i in the pure liquid or vapor states at the system temperature and pressure. Click here to enlarge imageįor ideal solutions for which Amagat’s Law of additive volumes holds, the fugacities iL and iV can be found with Equations 3 and 4, which are known as the Lewis and Randall Fugacity Rule.1Ĭombining Equations 2-4 gives the relationship in Equation 5 for ideal K values. An expression for Ki formulated in terms of the fugacity equality is Equation 2. The bar above f signifies value of fugacity in a mixture. The superscripts, V and L, indicate vapor and liquid phases, respectively, and the subscript i denotes a given component.
Depriester chart for equilibrium tv#
The equilibrium phase distribution ratio (K value) is defined in Equation 1 (see attached equation box).įor a system in equilibrium, TV = TL, PV = PL, and iV = iL Other methods require looking up charts or figures, where accuracy can be compromised. For preliminary design work and checking performance of existing columns, however, K values for multicomponent systems based on system pressure and temperature suffice.ĭepriester nomographs do not explain the basis how the K values are obtained.
Depriester chart for equilibrium software#
The K values thus obtained depend on pressure and temperature of the multicomponent system and not on concentration.Ĭoncentration-dependent K values can be calculated from empirical correlations involving lengthy calculations or from equations of state using commercial software because hand calculations are unwieldy. Depriester nomographs can be used to obtain estimates of K values. This article also explains the first principles for calculating K values from fugacity of real gases and liquids.Ī K value is required whenever one designs a distillation column or assesses the performance of an operating column. The proposed method, therefore, requires looking up only one chart-the generalized fugacity chart (which is a well-spaced-out chart and can be read accurately) and the rest of the calculations use equations, thus enabling rapid calculation. The method is based on the generalized fugacity charts of Lewis, Kay, and Newton, and uses reduced temperatures and pressures as parameters.Ī third-degree polynomial is prepared from Lu’s chart for calculating liquid molarity, which is used for calculating liquid phase fugacity from fugacity of saturated liquid. Post memes/jokes in /r/chemistrymemes and /r/chemistryjokes.A new method to estimate equilibrium phase distribution ratios (K values) improves on the shortcomings of older methods. Any such posts will be deleted.Īsk education and jobs questions in the current weekly topic. If you're looking for a more concentrated, advanced discussion of chemistry topics among professionals and grad students, check out /r/Chempros.īefore asking "What chemical is this?" see this chart. Click here for the OSHA chemical data site and here for a multicompany MSDS aggregate search. If you spill/injure yourself contact medical professionals and read the MSDS, do not post to this reddit. Yes links to blogs, images, videos, comics, and infographics are okay especially if they are on your personal website. No physorg, sciencedaily, or other press release aggregator spam!
Depriester chart for equilibrium license#
If a caption or explanation is included this helps, but please use your discretion.īefore asking about chemical drawing/illustration programs, look at your school's IT/software website and see if they provide an institutional license of ChemDraw (hint: if they have a chemistry department, they will) Likewise, simple pictures of uninteresting and garden variety chemistry-related things are not appreciated. No memes, rage comics, image macros, reaction gifs, or other "zero-content" material.
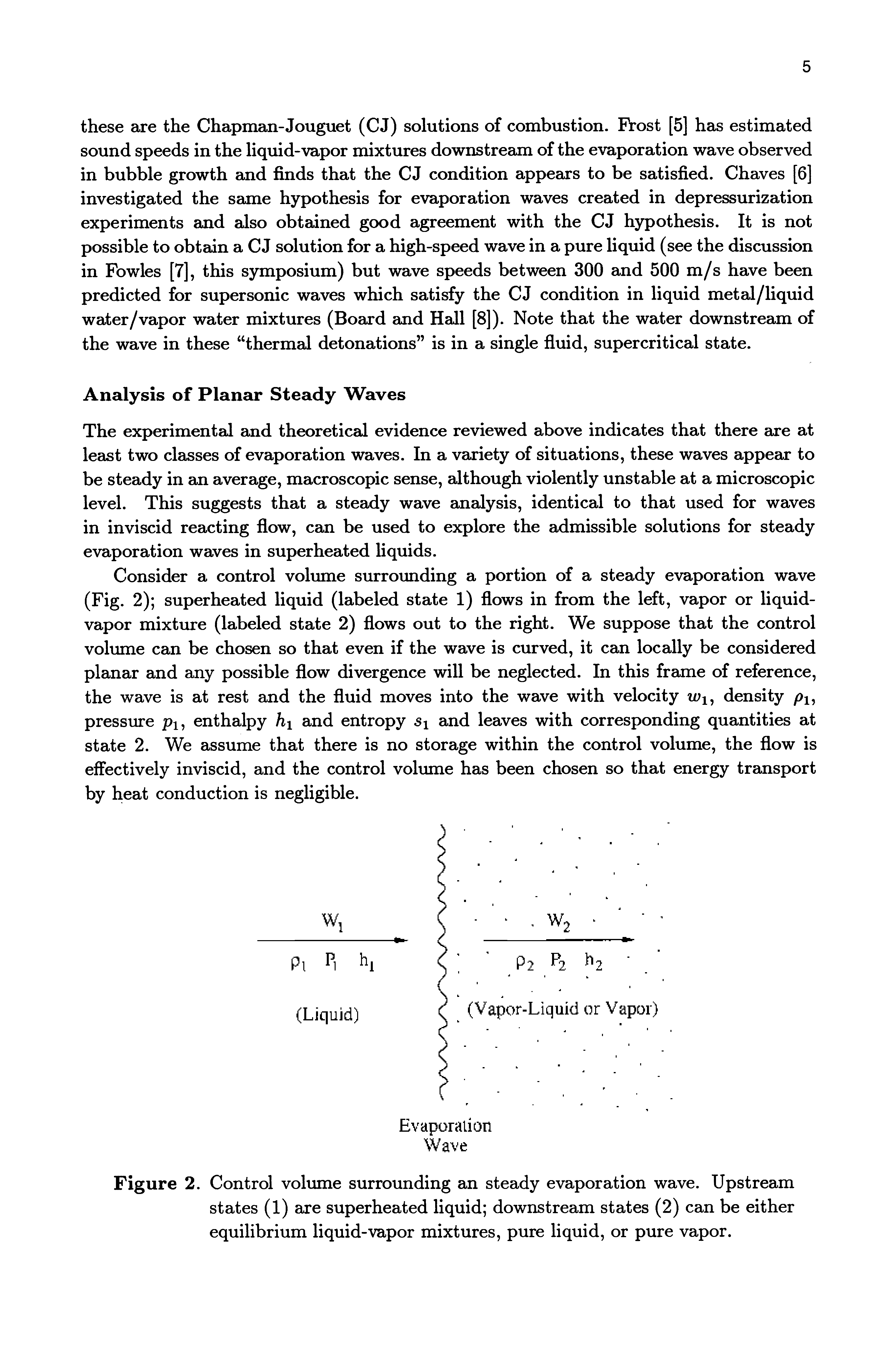
However, academic discussions on pharmaceutical chemistry and the science of explosives are permitted. Rules: Violating a rule will result in a ban.Īsk homework, exam, lab, and other undergraduate-level questions at ChemicalForums otherwise it will be deleted.ĭiscussions on illicit drug synthesis, bomb making, and other illegal activities are not allowed and will lead to a ban.
